Chemistry & Cleaning: What’s pH Got To Do With It?
People don’t roll out of bed one day and say, “I’m a cleaning expert!” What many people don’t realize is that a ton of science is involved in cleaning – especially if you want to do it right!
Basic chemistry principles are applied constantly while cleaning - both at home and professionally.
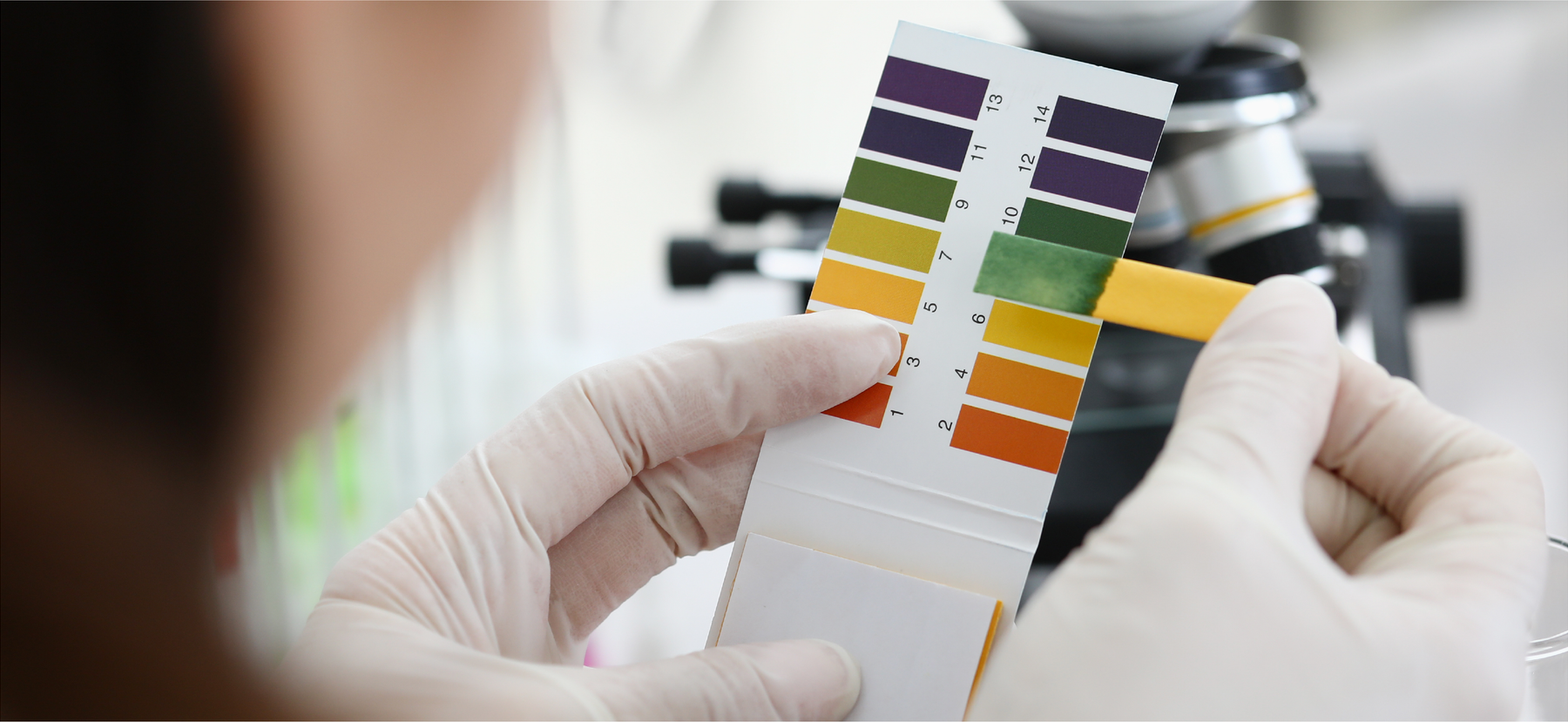
PH is important to keep in mind when cleaning, because keeping surfaces at a neutral pH is crucial to cleanliness, finish adhesion and shine. PH is measured by number values and ranges from acidic (0) to neutral (7) to alkaline (14). Neutral is valued at 7, anything below 7 is acid, and anything above 7 is base or alkaline. Here are a few common substances and their pH values.
So, why does pH matter? Most detergents are made up of alkaline materials in order to combat acidic soils left on a given surface. The alkaline detergent tackles the acidic soils from food remnants and other stains attempting to get the surface back to neutral. This is why measuring your detergents is so important. If you use too much detergent, you’ll find that the balance tips too far to the alkaline side causing sticky residue and dulling the surface. Not enough detergent and you’ll find it difficult to remove all the soils.
Descalers are acidic in order to combat alkaline mineral deposits left behind by hard water and other alkaline deposits.
So, simply stated:
- Bleach, ammonia and most degreasers are base or alkaline.
- Descaling cleaners, Muriatic Acid and vinegar are all commonly used acidic cleaners.
- Neutral cleaners are designed to simply use surfactants to disperse and remove dirt.
If you want to remove soils effectively, know your soils and know your surface make-up. Some surfaces can be damaged by high pH alkaline agents. Some can corrode if exposed to an agent that is highly acidic.
A great reason to use neutral pH cleaners (when you can) is that they generally are much safer and make compliance with OSHA’s new Globally Harmonized System requirements much easier. The Globally Harmonized System (GHS) requires that all commercial chemical labels display pictograms that offer info on the relative safety of the chemicals and the potential health hazards of the chemicals.

Check your product labels, measure everything exactly and if you don’t know for sure, check into it further. And of course, keep your pH levels balanced!



 Protect & Save with SuperFreak.
Protect & Save with SuperFreak.